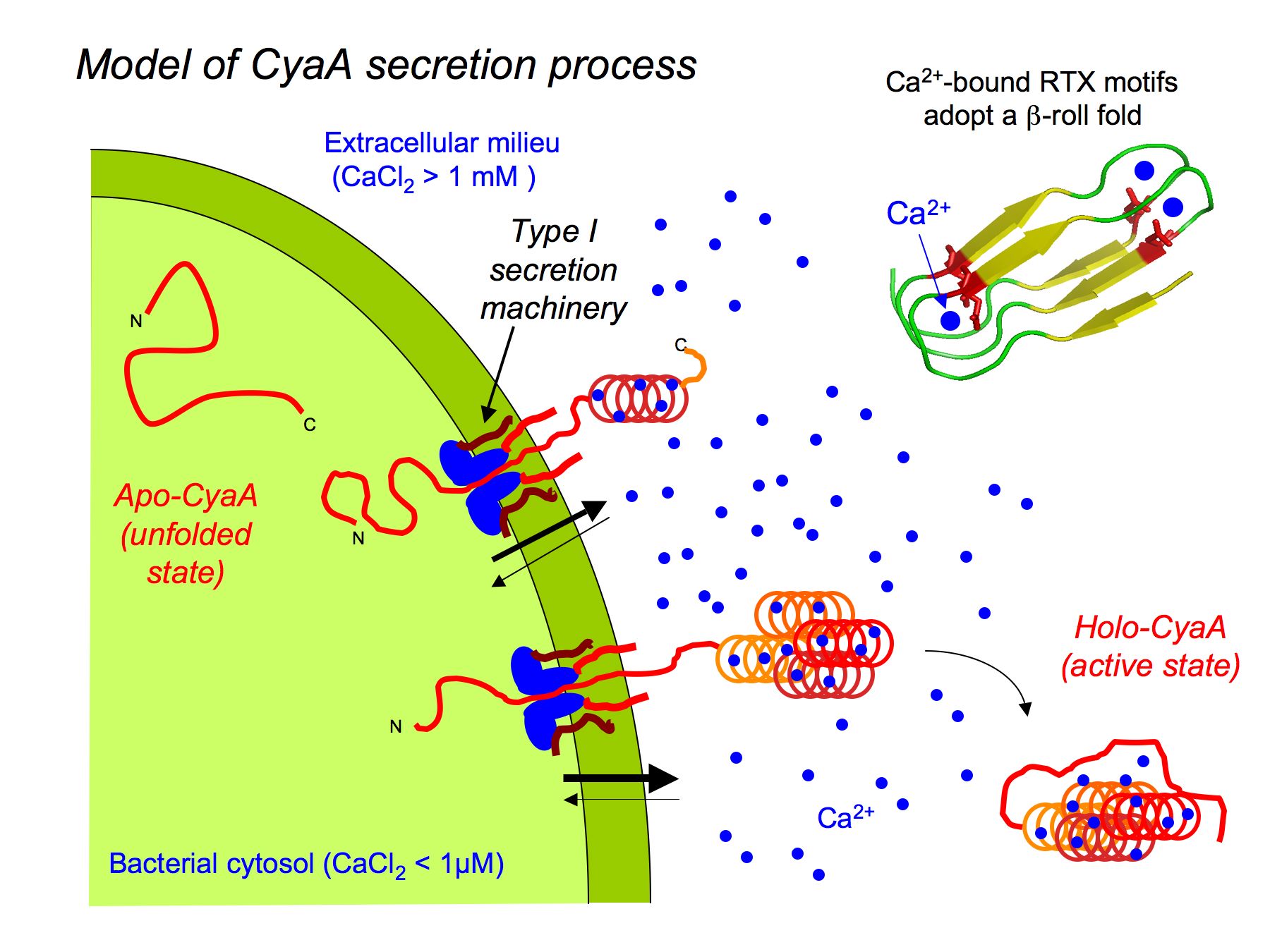
4 Schematic model for the secretion pathway of RTX toxins. Proposed... | Download Scientific Diagram

Identification of a Vibrio cholerae RTX toxin gene cluster that is tightly linked to the cholera toxin prophage | PNAS

In vivo covalent cross‐linking of cellular actin by the Vibrio cholerae RTX toxin | The EMBO Journal

RTX proteins: a highly diverse family secreted by a common mechanism - Linhartová - 2010 - FEMS Microbiology Reviews - Wiley Online Library

The RTX pore-forming toxin α-hemolysin of uropathogenic Escherichia coli: progress and perspectives. | Semantic Scholar

Bacterial RTX Toxins Allow Acute ATP Release from Human Erythrocytes Directly through the Toxin Pore* - Journal of Biological Chemistry

Almost half of the RTX domain is dispensable for complement receptor 3 binding and cell-invasive activity of the Bordetella adenylate cyclase toxin - Journal of Biological Chemistry

His-859 Is an Essential Residue for the Activity and pH Dependence of Escherichia coli RTX Toxin α-Hemolysin* - Journal of Biological Chemistry

Channel formation by RTX-toxins of pathogenic bacteria: Basis of their biological activity - ScienceDirect
Structural basis for antibody binding to adenylate cyclase toxin reveals RTX linkers as neutralization-sensitive epitopes | PLOS Pathogens
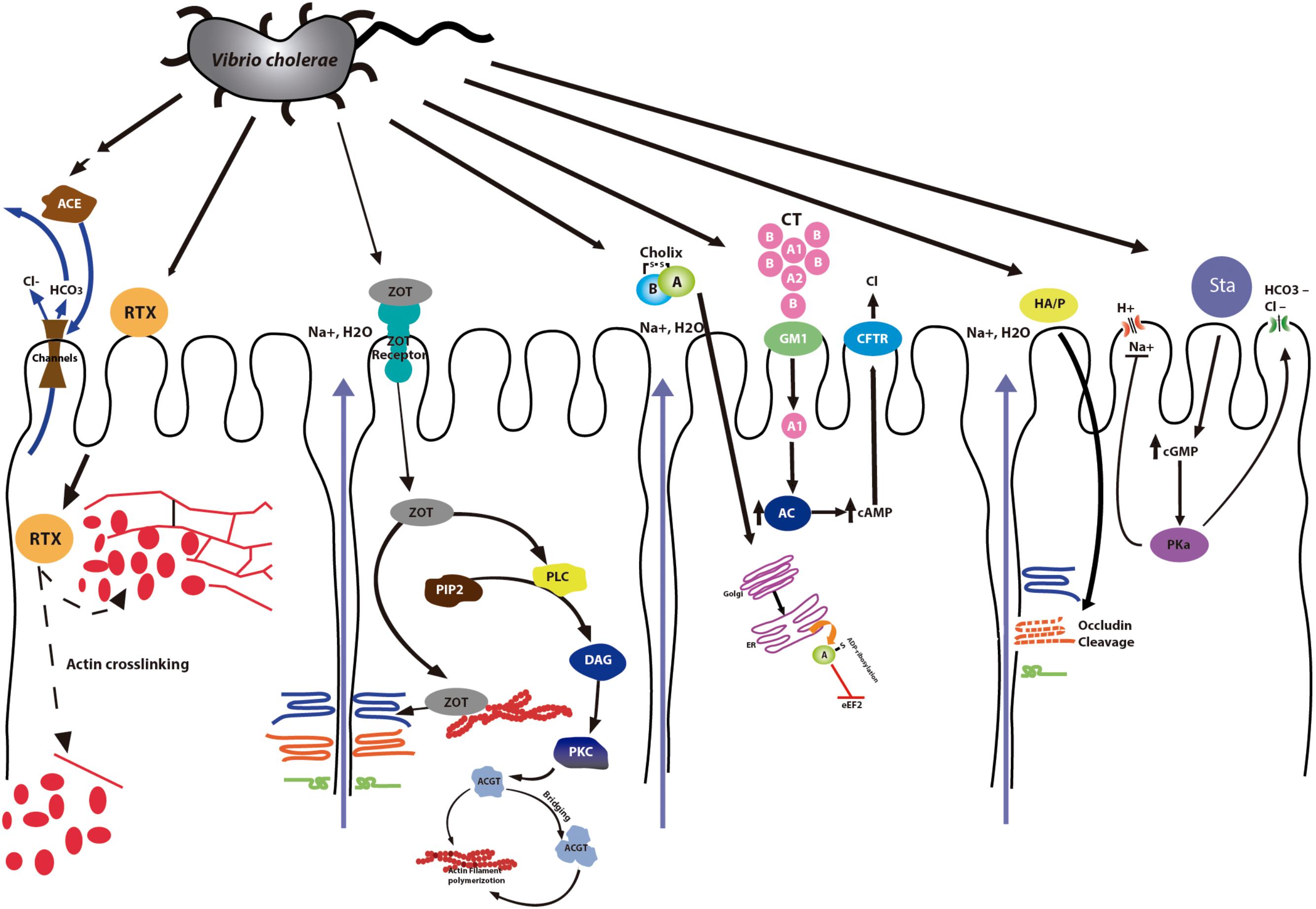
Frontiers | Accessory Toxins of Vibrio Pathogens and Their Role in Epithelial Disruption During Infection

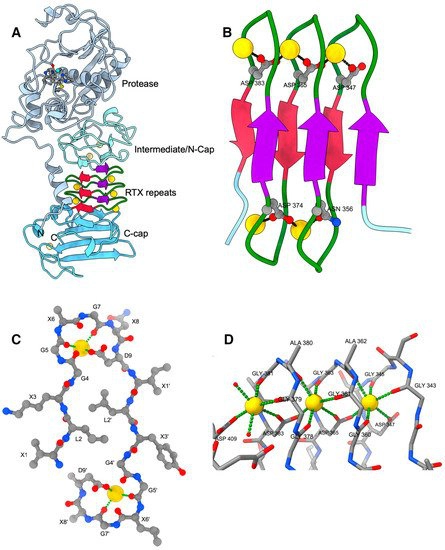
Toxins MDPI on Twitter: "Structure–Function Relationships of the Repeat Domains of #RTX Toxins https://t.co/JZ5sS9O77e #calcium; #proteinfolding; #tertiarystructure #mdpitoxins https://t.co/49AAZ478C5" / Twitter

Templated folding of the RTX domain of the bacterial toxin adenylate cyclase revealed by single molecule force spectroscopy | Nature Communications